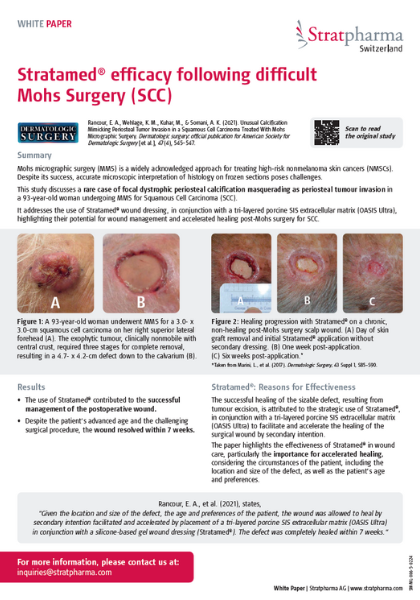
Stratamed allows the prevention of abnormal scarring to begin immediately after suturing or once a wound has occurred
Stratamed Clinical Efficacy
1. Stratamed® VS Topical Antibiotic (Neosporin® Original)
Benedetto, AV, et al. (2021). J Eur Acad Dermatol Venereol, 35(1) pp. 247-255.
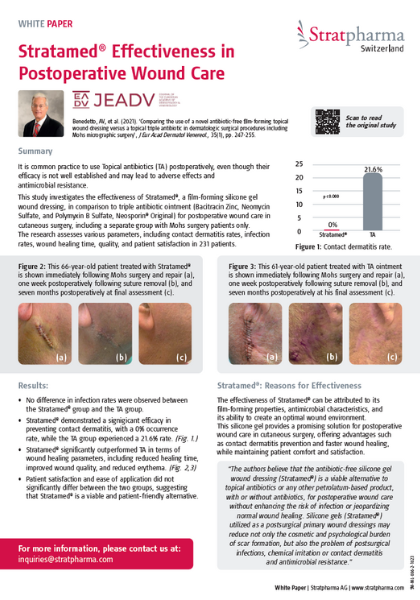
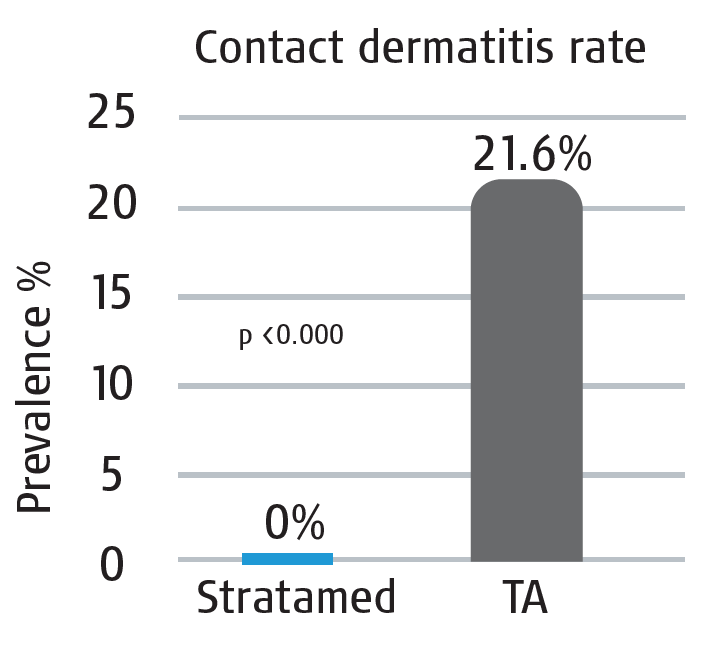
A 231-patient open-label, randomised, single-blinded study was conducted in the USA to compare Stratamed and a topical triple antibiotic ointment (Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate, Neosporin® Original) for the infection rate, prevalence of contact dermatitis and healing quality.
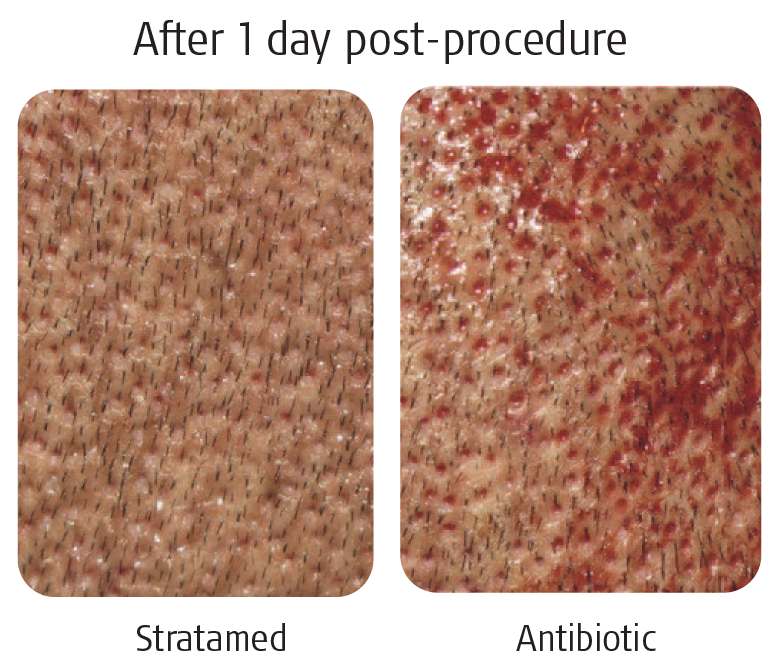
Results: Stratamed® demonstrated a significant efficacy in preventing contact dermatitis, with a 0% occurrence rate, while the triple Antibiotics (TA) group experienced a 21.6% rate.
- Stratamed, as an antibiotic-free gel, is a viable alternative to TA or any other petrolatum-based product, with or without antibiotics.
- Stratamed is as effective as TA in preventing infections without the risk for contact dermatitis.
- Stratamed promotes faster healing, reduced erythema and better quality of the newly formed tissue.

2. Stratamed VS Topical Antibiotic (Bacitracin®)
James, IB, et al. (2022). Aesthetic Surgery Journal Open Forum, 4, pp. 1-8.
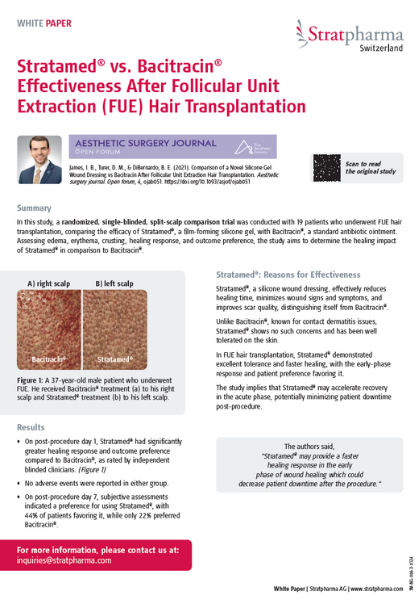
A 19-patient randomised, single-blinded, split-scalp trial was conducted in the USA to compare Stratamed and topical antibiotic (Bacitracin Zinc, Bacitracin®) assessing the edema, erythema, crusting, healing response and outcome preference following FUE (Follicular Unit Extraction) hair transplantation.
Results: Stratamed is as effective as topical antibiotics in reducing the inflammation (edema, erythema and crusting).
- Faster healing response was observed as early as day 1 in the Stratamed group.
- Stratamed avoids concerns of promoting bacterial resistance or significantly altering the skin microbiome.
3. Stratamed treatment to Non-Healing Burn Wound
Lucattelli, E., et al. (2021). Annals of Burns and Fire Disasters, 34(1), pp. 53-57.
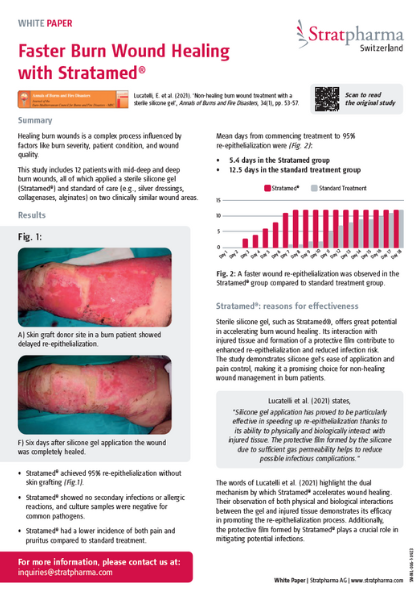
Study with 12 patients with mid-deep and deep burn wounds, all of which applied Stratamed® and standard of care (e.g., silver dressings, collagenases, alginates) on two clinically similar wound areas.
Results:
Mean days from commencing treatment to 95% re-epithelialization were:
– 5.4 days in the Stratamed group
– 12.5 days in the standard treatment group
- Stratamed showed no secondary infections or allergic reactions, and culture samples were negative for common pathogens.
- Stratamed had a lower incidence of both pain and pruritus compared to standard treatment.
- Stratamed speeds up the healing process - 2 times faster wound healing compared to standard treatment
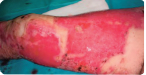
Start of treatment with Stratamed
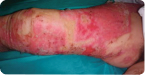
After 6 days of treatment with Stratamed
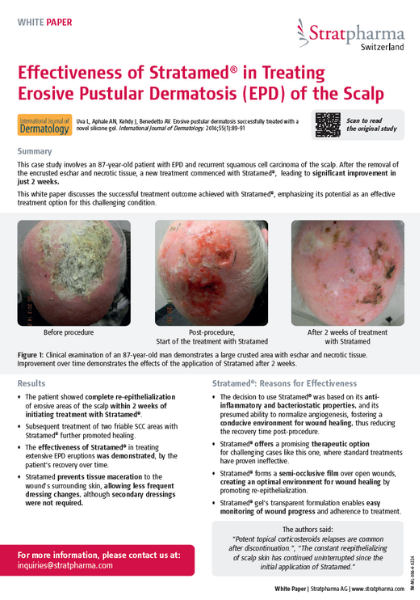
4. Stratamed®- Successful treatment of Chronic Scalp Wounds – Monk
Monk, E., et al. (2014). ‘Successful Treatment of Nonhealing Scalp Wounds Using a Silicone Gel’, Dermatologic Surgery, 40(1), pp. 76–79.
This study showcases the efficacy of Stratamed in treating non-healing scalp wounds, through an in-depth examination of four case studies involving elderly patients who underwent surgical procedures for scalp carcinomas.
Despite initial treatments, including topical ointments and electrodesiccation with curettage, the wounds remained nonhealing.
Results:
- Significant improvements were observed in all cases, after the application of Stratamed directly onto the wound, as monotherapy without a secondary dressing.
- The results demonstrated improved wound healing within a 2-month period following the inciting event.
Start of treatment with Stratamed
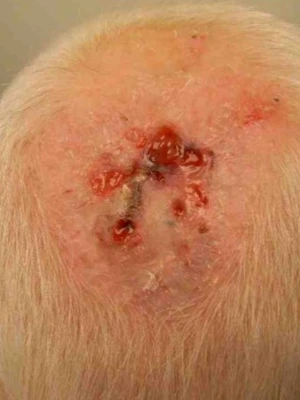
2 Years after surgery, start of treatment with Stratamed
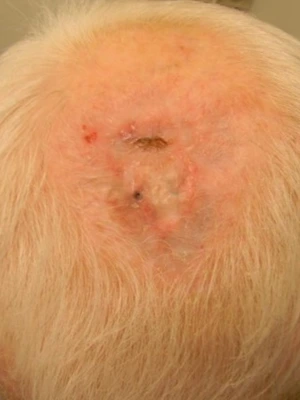
After 5 weeks of treatment
List of Major Clinical Publications
The following studies have been published in international journals confirming the unique and novel nature of Stratamed.

Cubiró, RX, et al. (2019). ‘Efficacy of an Innovative Film-Forming Wound Dressing on Open Wounds’, SKINmed, 17(5), pp. 298-304.

Uva, L. et al. (2016).‘Erosive pustular dermatosis successfully treated with a novel silicone gel’, International Journal of Dermatology, 55(1), pp. 89-91.

Monk, EC, et al. (2014). ‘Successful Treatment of Nonhealing Scalp Wounds Using a Silicone Gel’, Dermatologic Surgery, 40(1), pp. 76-79

Sandhofer, M., Schauer, P. (2012). ‘The Safety, Efficacy, and Tolerability of a Novel Silicone Gel Dressing Following Dermatological Surgery’, SKINmed, 10(6), S1-S7.
Whitepapers
Discuss with our medical team
If you would like to take part in one of our clinical trials, or if you have any other questions, please don’t hesitate to contact us.
Our medical team will be delighted to answer your questions.